Helium is the least reactive element in this group, creating no stable compounds. Noble gases are the least reactive elements. 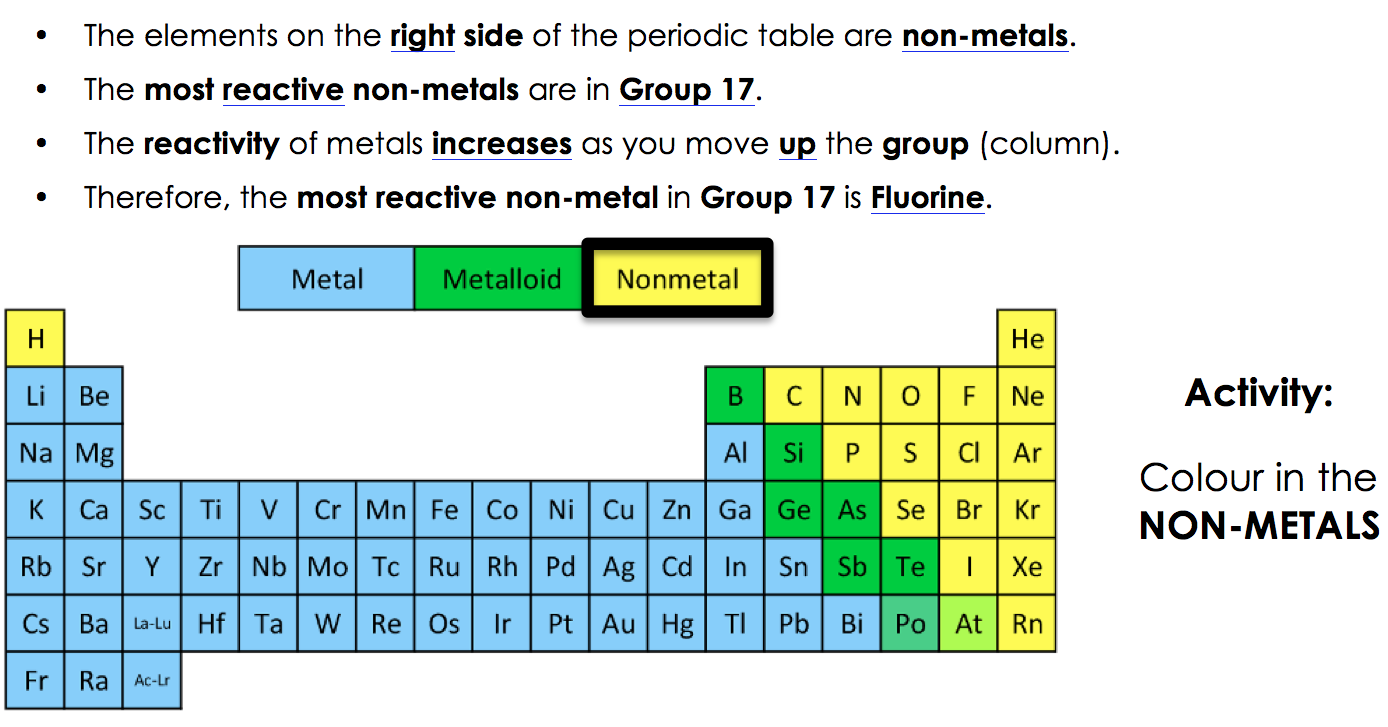
Cesium, which lies right above francium on the periodic table, is the most reactive metal with a stable isotope. However, francium is a radioactive element that is only present in tiny levels. The most reactive metal is francium, which is also the final alkali metal (and the most expensive element). Halogens, alkali metals, and alkaline earth metals are all extremely reactive.įluorine, the first element in the halogen group, is the most reactive. These elements are found in the periodic table’s top right and lower left corners, as well as in certain element groups. Highly electropositive and highly electronegative materials both have a great proclivity to react. The periodic table’s arrangement of elements allows for reactivity predictions. Reactivity may also be defined as the scientific study of chemical processes and their kinetics. Temperature increases the amount of energy available for a chemical reaction, making it more likely. The temperature has an effect on reactivity. 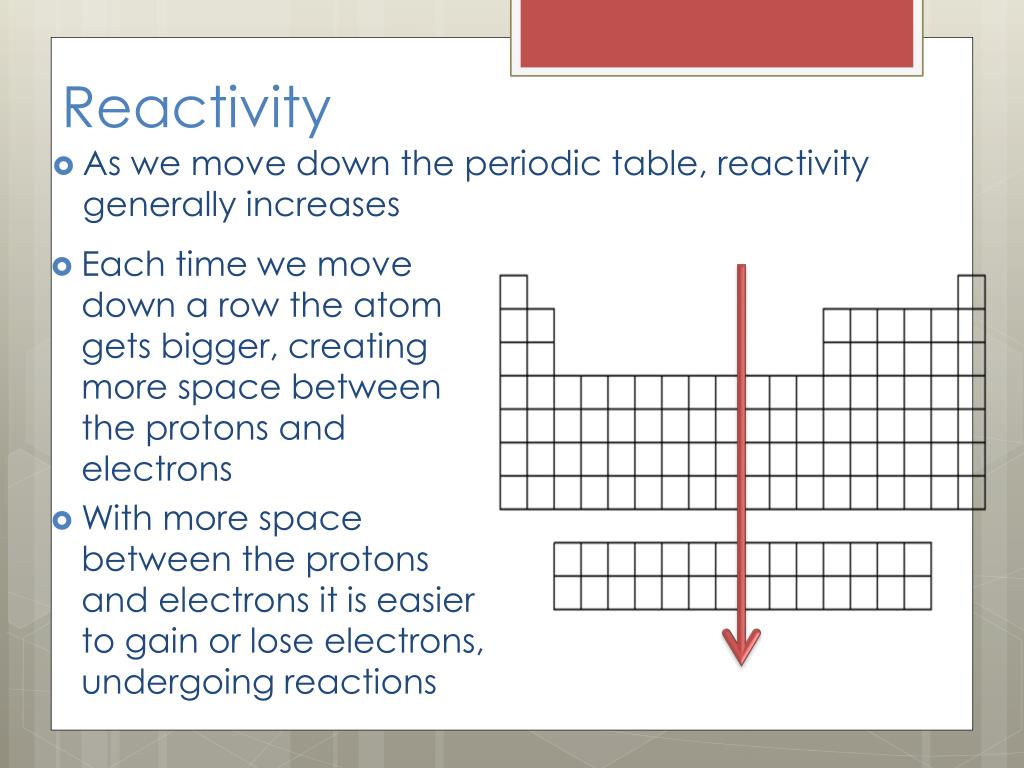
They often burn in both water and oxygen in the air. The most reactive elements and compounds have the potential to spontaneously or explosively ignite. The reaction might include the chemical alone or in combination with other atoms or molecules, and it is usually followed by the release of energy. Reactivity is a measure of how easily a material conducts a chemical reaction in chemistry.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
